Attillaps Pharmaceuticals · Dermatology & Ophthalmology
The AChM Platform Acetylcholine Modulators that kill Demodex
Targeting the unified biological driver of rosacea, blepharitis, and the cutaneous autoimmune spectrum.
Section 1
The Demodex–Immune Dysregulation Spectrum
One parasite. Multiple antigens. A spectrum of inflammatory disease from rosacea to lupus — and a 70-year mystery finally explained.
Every adult human carries Demodex mites. DNA-based detection finds them on 100% of adults over 18. For most people the immune system silently tolerates them. But for millions of patients, a breakdown in immune regulation allows the mite’s primary structural component — chitin — to trigger a chronic inflammatory cascade. What medicine calls “rosacea,” “cutaneous lupus,” or “blepharitis” may all be expressions of the same immunological event at different severity levels.
Same root cause — Demodex antigen hypersensitivity — expressed at increasing immune dysfunction severity
The Demodex Immune Cascade
Each link in this chain is independently confirmed in published literature. This is not a hypothesis about the cascade — it is a hypothesis about what HCQ is actually interrupting.
Is Rosacea a Systemic Autoimmune Disease?
The Venn diagram below illustrates the substantial overlap between rosacea and lupus — sharing cytokines, immune pathways, and critically, the same treatments. The two conditions differ not in kind but in severity, consistent with a single shared driver: Demodex-mediated chitin immune dysregulation expressed at different points on the spectrum.
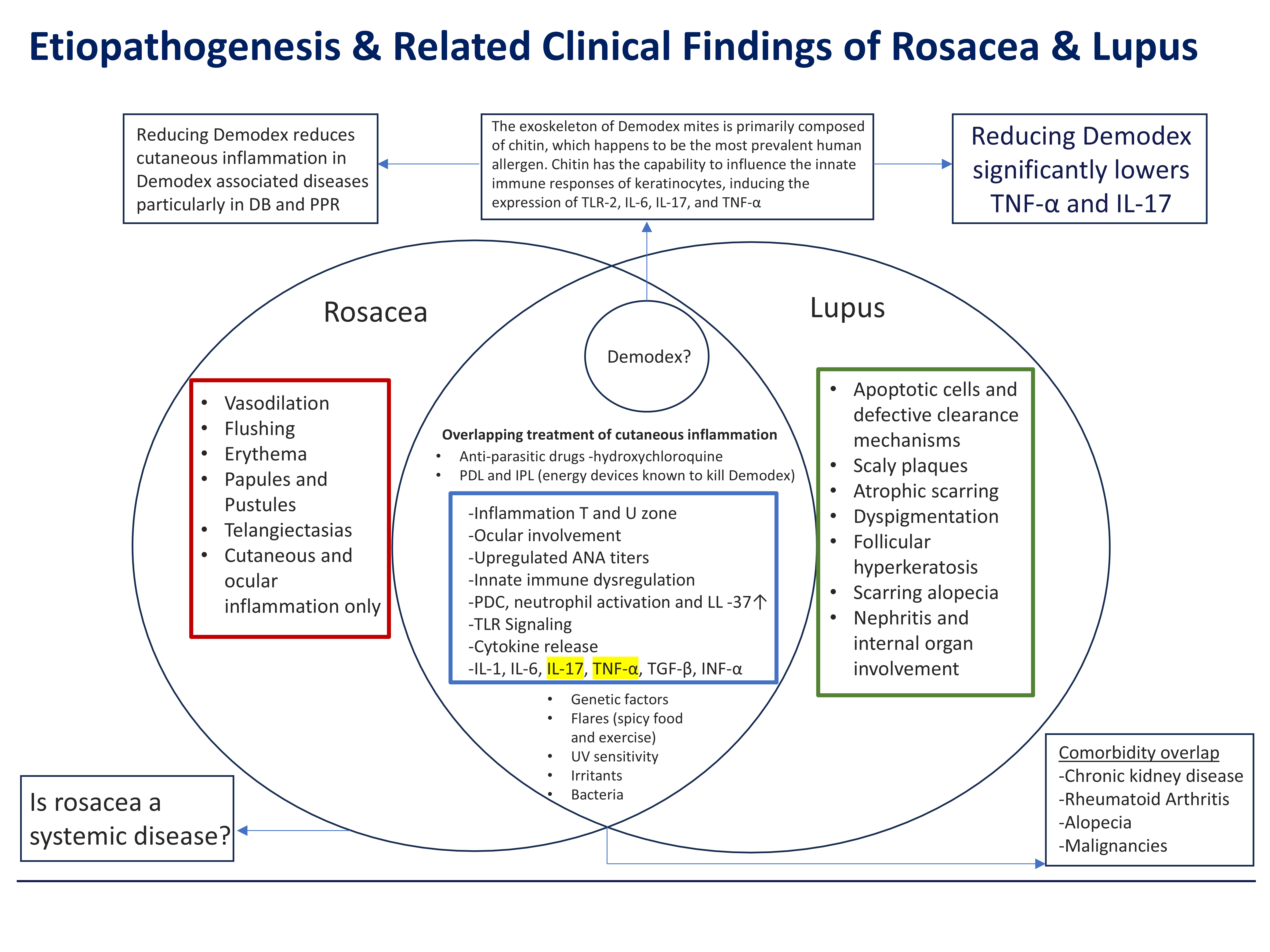
Figure 1. Etiopathogenesis and Related Clinical Findings of Rosacea and Lupus. The Venn diagram illustrates the substantial overlap between rosacea (left) and lupus (right), with Demodex proposed as the central unifying driver. Overlapping features include inflammation in the T and U zones, ocular involvement, upregulated ANA titers, innate immune dysregulation, PDC and neutrophil activation, TLR signaling, and shared cytokines (IL-1, IL-6, IL-17, TNF-α, TGF-β, INF-α). Overlapping treatments — anti-parasitic drugs (hydroxychloroquine) and energy devices (PDL, IPL) known to kill Demodex — further support the unified etiology. Rosacea-only features (red box) represent localized cutaneous/ocular inflammation; lupus-only features (green box) represent systemic organ involvement — consistent with differing severity of the same Demodex chitin-driven immune dysregulation spectrum. Adapted from Dursun et al. Dermatologic Therapy 2020.
Hydroxychloroquine has been used to treat lupus, RA, and 8+ autoimmune diseases for 70 years with no definitive mechanistic explanation. Attillaps proposes the answer: HCQ kills Demodex in the pilosebaceous unit using the same anti-parasitic mechanism it uses against Plasmodium. The immunological effects attributed to HCQ are not primary drug actions — they are downstream readouts of an immune system that no longer has a parasite driving its activation.
Section 2
Hydroxychloroquine: The Established Standard of Care
HCQ is not a competitor to the AChM platform — it is the proof-of-concept reference compound and the current standard of care.
Ex Vivo Kill Data: Chloroquine 10% Kaplan-Meier Survival Analysis
Attillaps has confirmed statistically significant chloroquine miticidal activity against Demodex in standardised ex vivo LT50 assays. Chloroquine diphosphate is the validated aqueous surrogate for HCQ in water-based assay conditions, by virtue of structural equivalence: the two compounds are congeners differing by a single hydroxyl substituent on the ethyl side chain, conferring shared pharmacological mechanisms. The basis for surrogate selection is structural and mechanistic, not solubility-driven — the hydroxyl group in HCQ increases rather than decreases aqueous solubility relative to chloroquine. Permutation testing on the primary LT50 endpoint confirms the treatment effect at p = 0.0012.
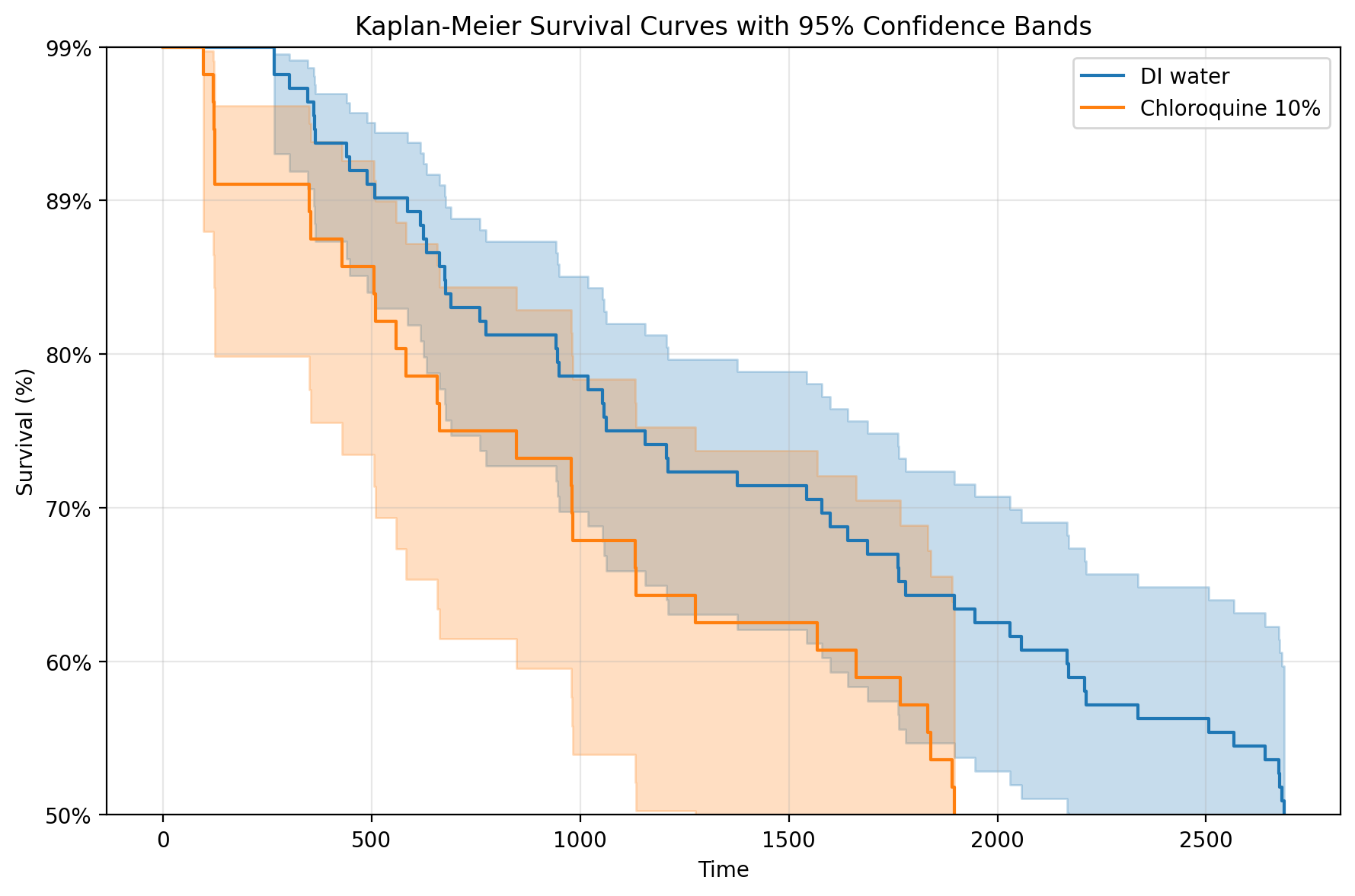
Figure 2. Kaplan-Meier survival analysis. Orange: chloroquine-treated mites. Blue: water vehicle control. Shaded bands: 95% confidence intervals. Attillaps internal ex vivo assay data, N > 50 mites per arm.
The water-based assay has no lipid partitioning or follicular depot effect. Chloroquine’s lipophilicity (logP ≈ 4.6) predicts preferential partitioning into follicular sebum, concentrating the effective mite-surface dose well above the nominal 10% applied concentration. The in vitro LT50 of 1,896 minutes is a conservative floor — the true in vivo LT50 in a sebum-formulated preparation would be expected to be substantially shorter.
HCQ vs. AH-300 (AChM Platform) — Head-to-Head Comparison
| Parameter | Hydroxychloroquine (HCQ) — Systemic Standard | AH-300 (AChM) — Attillaps Platform |
|---|---|---|
| Molecular Weight | Note335.9 Da — Below 500 Da rule | Advantage<500 Da — Full follicular penetration |
| Kill Mechanism | SingleLysosomal pH disruption — anti-parasitic · systemic delivery only | Multi-targetMultiple cholinergic pathways · confirmed contact kill |
| Direct Contact Kill | IndirectConfirmed ex vivo — but only via slow systemic depot | LT50 <12 hrsAll compounds confirmed · rapid contact activity |
| Sebum Drug Level | SlowBuilds over weeks — depot-dependent, months to therapeutic sebum levels | ImmediateTopical application delivers drug directly to site of action |
| Resistance Risk | LowSingle mechanism — decades of use, no known Demodex resistance | Very LowMultiple independent neuro-targets |
| Route of Administration | Oral onlyRetinal toxicity risk with long-term use | TopicalNo systemic exposure · known safety via 505(b)(2) path |
| Indications | Lupus · RA · Malaria — off-label for some autoimmune, not approved for rosacea/blepharitis | Full SpectrumRosacea · Lupus · Blepharitis · Acne · RA · Full DCIDS |
Patients with SLE, RA, and autoimmune conditions are already receiving HCQ as standard of care. Attillaps clinical studies will evaluate AChM compounds added to standard HCQ therapy — measuring whether topical Demodex eradication at the primary site of antigen production produces incremental immunological normalization over and above what systemic HCQ alone achieves.
Section 3
The Downstream Measurement Hypothesis
A reframing of 70 years of HCQ mechanistic research — and why it matters for the entire autoimmune therapeutic landscape.
Section 4
Platform Advantages
The AChM platform is the first integrated IP estate targeting the root cause of Demodex-driven inflammatory disease across dermatology, ophthalmology, and rheumatology.
Contact Us to Learn More
The first integrated IP estate targeting the root cause of Demodex-driven inflammatory disease — across dermatology, ophthalmology, and rheumatology.